Bracco Diagnostics' MultiHance Contrast Agent Earns Expanded Labeling for Pediatric MRI
Bracco Diagnostics Inc. announced the labeling of its contrast agent MultiHance has obtained U.S. Food and Drug Administration (FDA) approval for an extension to include magnetic resonance imaging (MRI) of the central nervous system (CNS) in pediatric patients younger than 2 years of age (including term neonates). The agent may now be used in this patient population to visualize lesions with abnormal blood-brain barrier or abnormal vascularity of the brain, spine and associated tissues.

PDF) The utility of gadoteric acid in contrast-enhanced MRI: A review

Magnetic Resonance Angiography of the Thoracic Vasculature: Technique and Applications - Ludwig - 2020 - Journal of Magnetic Resonance Imaging - Wiley Online Library

Amidst Increase in MRI Procedures, New Survey Finds 55% of Radiologists Have Concerns About Contrast Agent Availability

Contrast-enhanced Magnetic Resonance Imaging in Pediatric Patients: Review and Recommendations for Current Practice - Ravi Bhargava, Gabriele Hahn, Wolfgang Hirsch, Myung-Joon Kim, Hans-Joachim Mentzel, Øystein E. Olsen, Eira Stokland, Fabio Triulzi, Elida

A liposomal Gd contrast agent does not cross the mouse placental barrier

Contrast-enhanced Magnetic Resonance Imaging in Pediatric Patients: Review and Recommendations for Current Practice - Ravi Bhargava, Gabriele Hahn, Wolfgang Hirsch, Myung-Joon Kim, Hans-Joachim Mentzel, Øystein E. Olsen, Eira Stokland, Fabio Triulzi, Elida
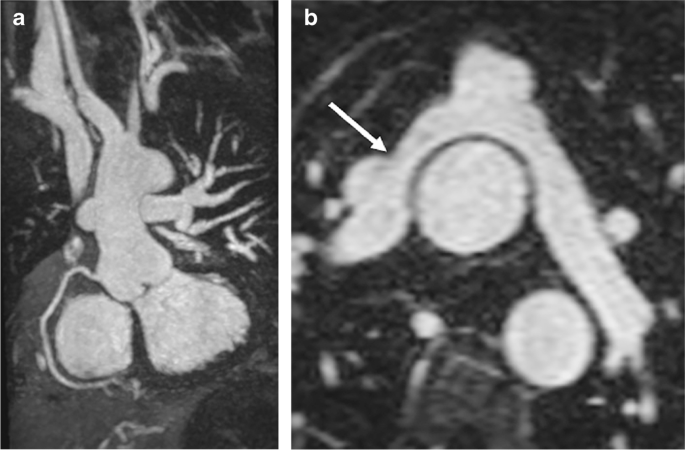
Pediatric magnetic resonance angiography: to contrast or not to contrast

Bracco Diagnostics' MultiHance Contrast Agent Earns Expanded Labeling for Pediatric MRI

Use of Eovist in Pediatric Patients: Pearls and Pitfalls - ScienceDirect

X-ray-computed tomography contrast agents. - Abstract - Europe PMC
Bracco Launches CT Exprès 3-D Contrast Media Delivery System at RSNA 2016

Use of Eovist in Pediatric Patients: Pearls and Pitfalls - ScienceDirect