
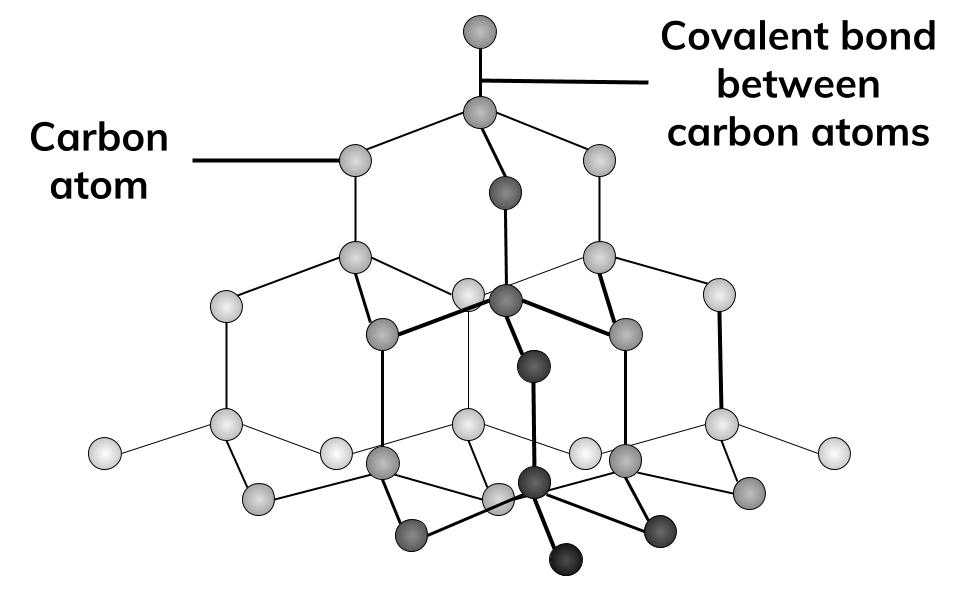
Does The Difference In Structure Make Graphite Soft But Diamond Hard?
At first, this question might seem odd to many people. Diamond and graphite… doesn’t sound like a particularly sensible combination. Diamond and gold, or diamond and sapphire would make more sense, right?
Science gets a bad rep for being boring, difficult and for very smart people (see: nerds). But that doesn't have to be the case. At ScienceABC we believe that if science is communicated simply and made relatable and fun, it can be for everyone. As Micheal Jackson said best, as simple as ABC. So far, we've tackled complex and vast topics like Einstein's Time Dilation, the Immune System, Evolution, Quantum Entanglement, while also asking the questions that nag us at night like Is the science in the movies accurate?, Is it possible to have insects the size of humans?, and Why can I hear a thundering sound in my ear?. So, join us for some simple science with a whole lot of fun thrown in! Subscribe to us for your regular dose of science and don't forget to share our work if you liked it.

Let's learn about diamond

Why is diamond hard and graphite brittle while both are made up of

Diamond Graphite

Two-Layer Graphene becomes a Diamond-Hard Material on Impact which

Graphite properties - Chemical properties - Definition, Fromulas

The Power in Your Pencil. Graphite, the black sheep of the carbon

Allotropes of carbon, Feature

Comparison of some properties of the two renowned allotropes of
1:50 explain how the structures of diamond, graphite and C60

Does The Difference In Structure Make Graphite Soft But Diamond Hard?

Minerals. - ppt video online download

Carbon lattices - VCE Chemistry
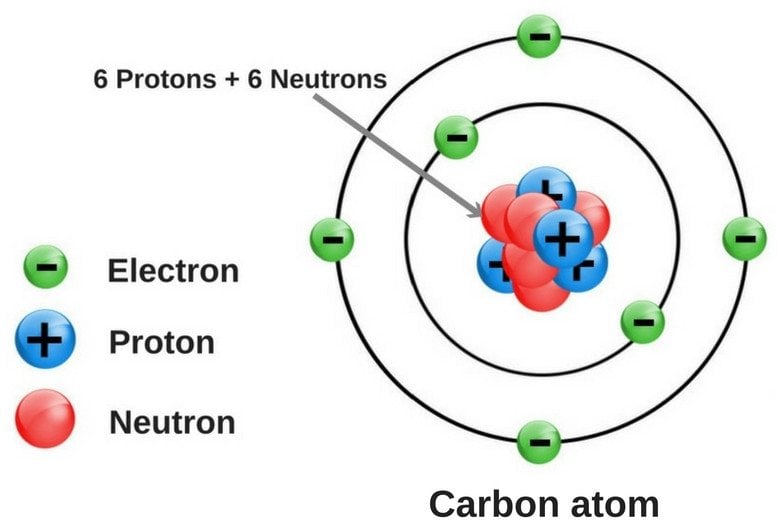
Does The Difference In Structure Make Graphite Soft But Diamond Hard?
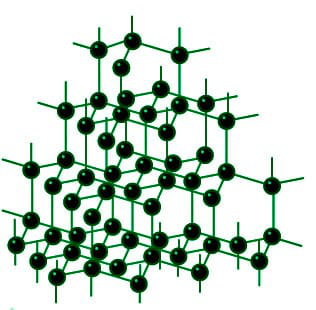
Diamond and Graphite - Structure, Uses, Properties, Applications
Why is graphite soft and diamond hard? - Quora









