.jpg)
FDA Alerts

Indian CRO hit with FDA warning letter over two specific violations

Ravin Ratan on X: Nirogacestat is approved by the FDA for

FDA Alerts Providers to COVID-19 Rapid Test Recall

FDA updates Ozempic label to include warning for intestinal blockage, hypoglycemia risk

Paul's FDA Alerts, Published Postman Templates

U.S. FDA on X: FDA alerts consumers of nationwide voluntary

FDA Alerts Device Manufacturers to GUDID Data Mismatch Issues

Accurate Seizure Data for Doctors, FDA-cleared Monitoring and Alerting, Embrace2
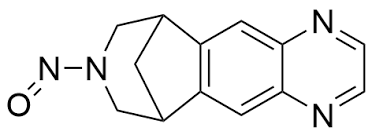
FDA alerts to a voluntary recall of varenicline - What do we know
FDA Alerts of Voluntary Strawberry Recall Due to Hepatitis A

FDA alerts health providers about other drugs from meningitis

FDA alerts public of unsafe Hydrocortisone injection

Recalls, Market Withdrawals, & Safety Alerts









