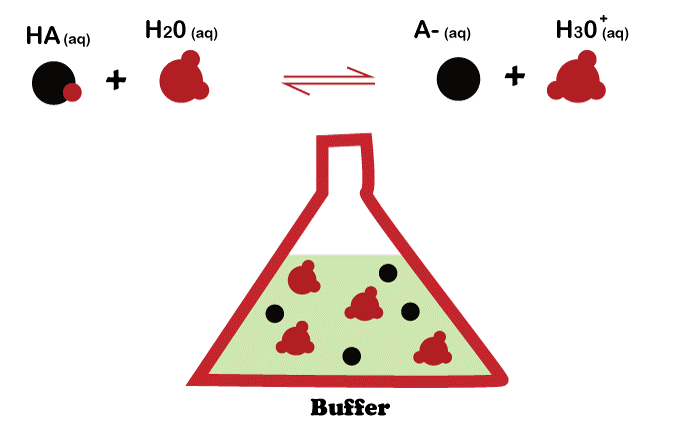
Buffer Solutions - Chemistry Steps
A buffer is a solution that contains either a weak acid with its salt or a weak base with its salt to maintain the pH of the solution.
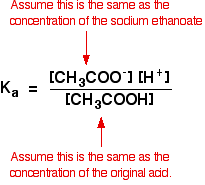
buffer solutions

Buffer Systems
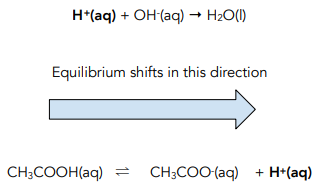
Acids and Bases - Buffer Action (A-Level Chemistry) - Study Mind

Buffer Solutions

Buffers: Common Ion Effect, Henderson-Hasselbalch Equation, and Buffer Capacity - Concept, Lab: Chemistry
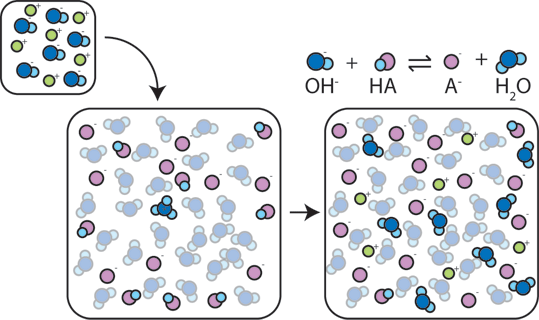
How Buffers Work
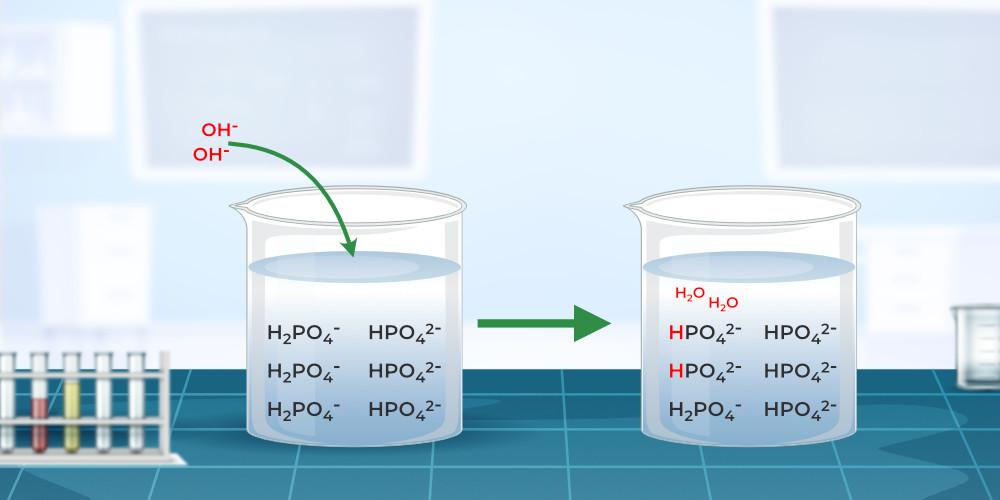
Buffer Solution - Definition, Types, Formula, Examples, and FAQs

Buffers 2 - Acid Base Equilibria - MCAT Content
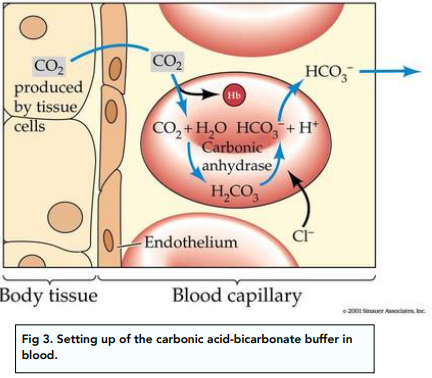
Acids and Bases - Buffer Action (A-Level Chemistry) - Study Mind

Chemistry Steps (@ChemistrySteps) / X

Solved You work in a chemistry lab, and are asked to prepare

Buffer solution - Wikipedia

Buffer Solution - Acidic and Basic Buffers, Preparations, Examples
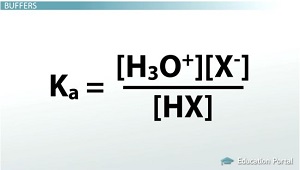
How To Calculate the PH of a Buffer Solution, Equation & Example - Video & Lesson Transcript