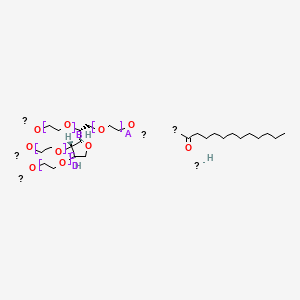
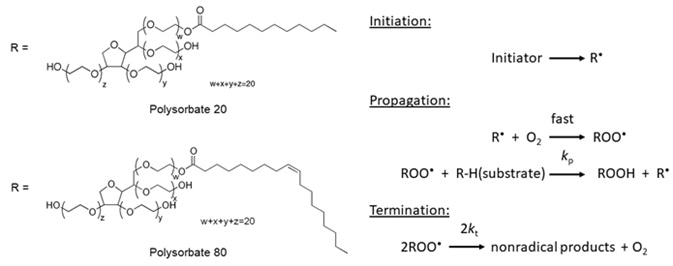
Polysorbates 20 and 80 Used in the Formulation of Protein

Quantitative Analysis of Polysorbate 20/80 in Protein-Based Biopharmaceuticals Using A One-Pot RPLC-MS Based Platform Method

WO2011089062A2 - Pharmaceutical formulation for proteins - Google Patents
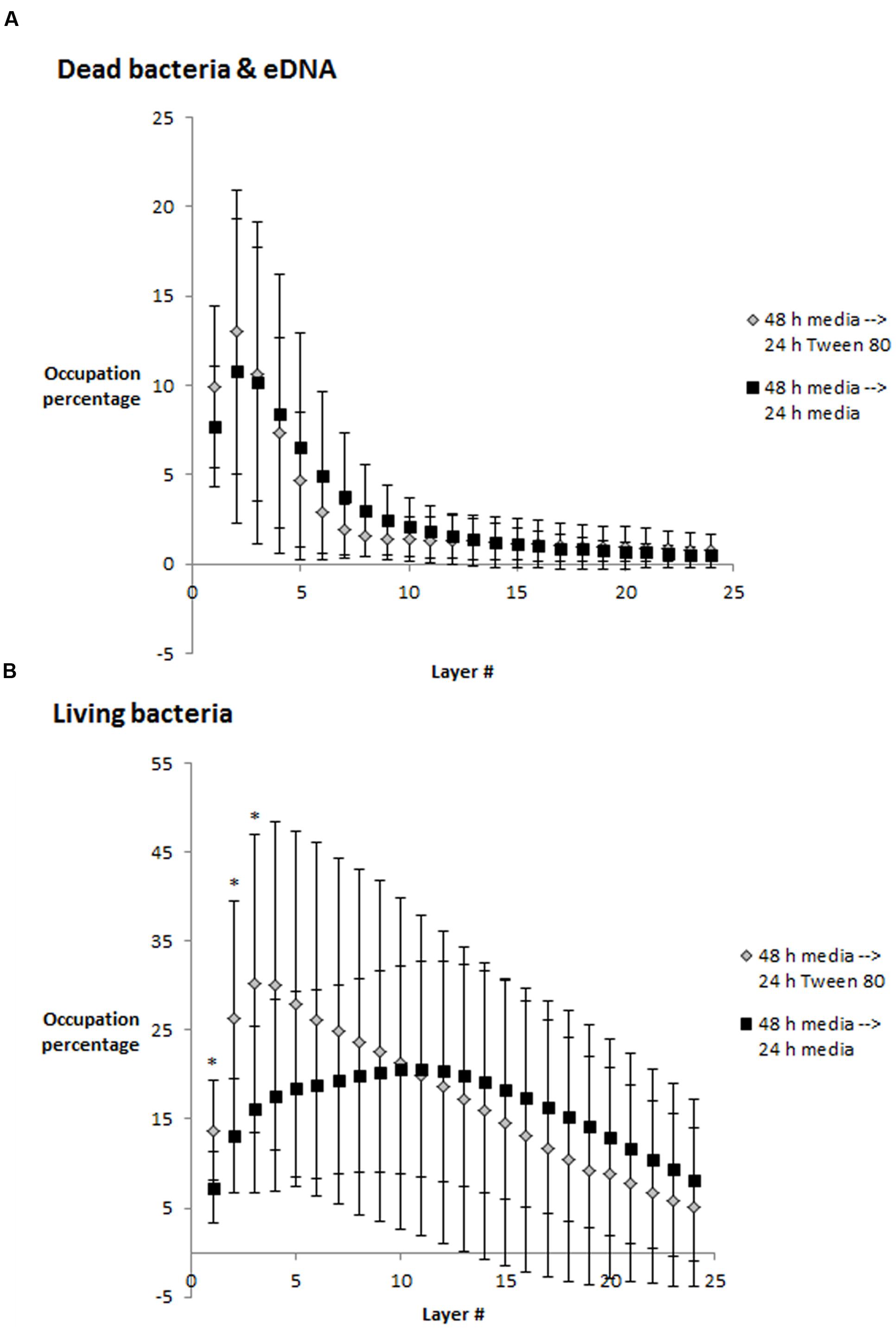
Frontiers Effects of Tween 80 on Growth and Biofilm Formation in Laboratory Media

WO2021007504A2 - Methods and compositions comprising reduced level of host cell proteins - Google Patents
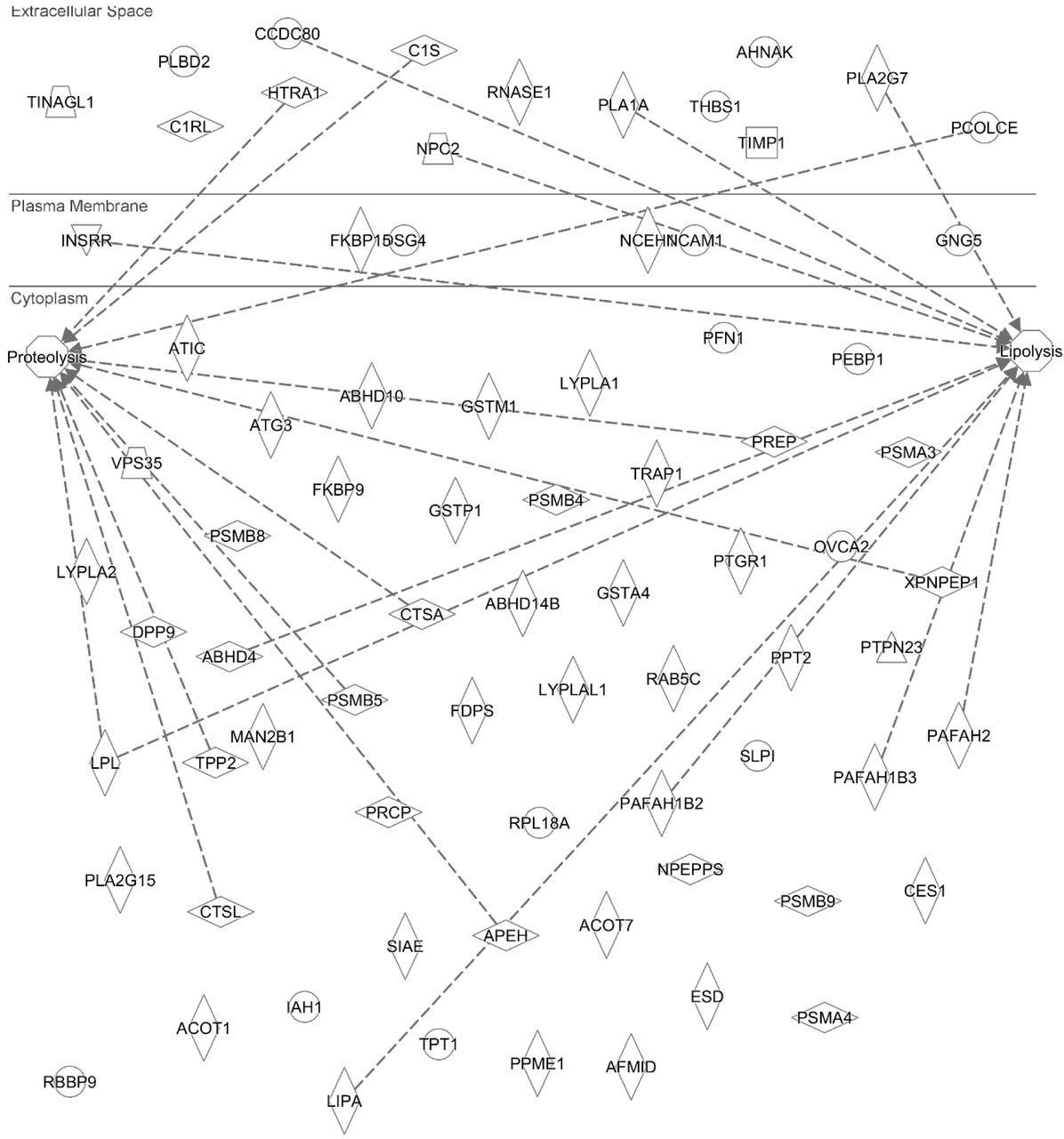
Profiling Active Enzymes for Polysorbate Degradation in Biotherapeutics by Activity-Based Protein Profiling
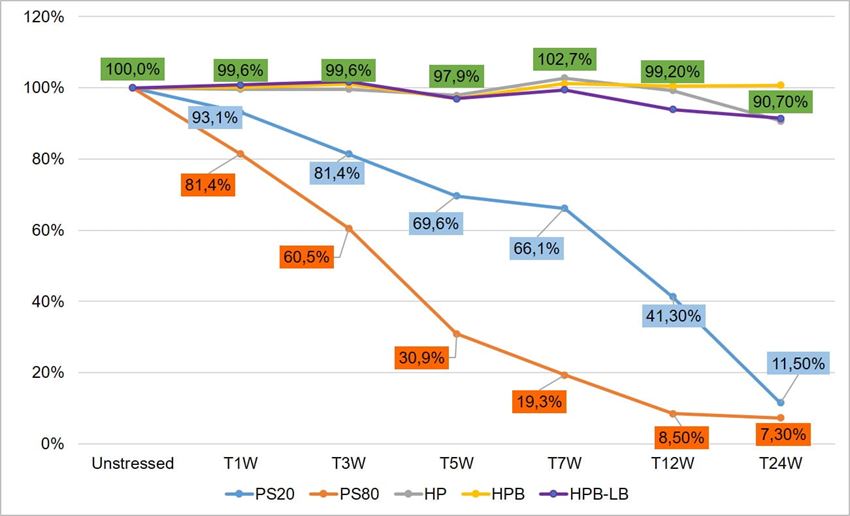
Polysorbates versus Hydroxypropyl Beta-Cyclodextrin: Comparative Study on Excipient Stability and Stabilization Benefits on Monoclonal Antibodies
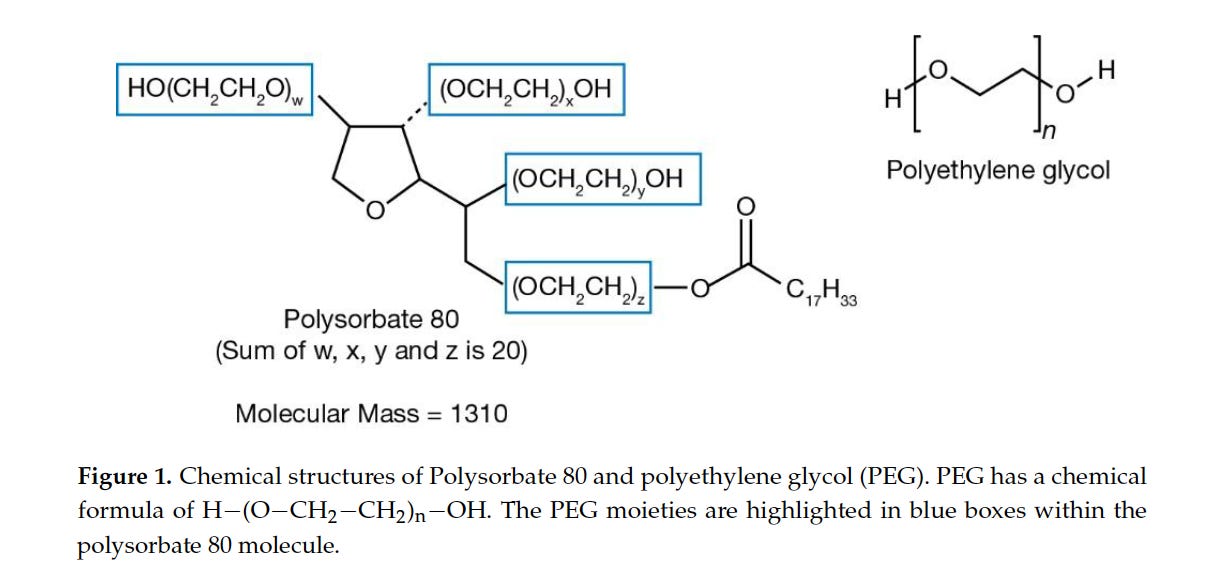
Polysorbate 80 in Covid19 Jabs a Major Hazard
What's the Difference Between Tween 20 and Tween 80?

Effect of Polysorbate 20 and Polysorbate 80 on the Higher-Order Structure of a Monoclonal Antibody and Its Fab and Fc Fragments Probed Using 2D Nuclear Magnetic Resonance Spectroscopy. - Abstract - Europe PMC
View of ANALYSIS OF POLYSORBATE 80 SOLUTION STABILITY UNDER STRESS CONDITIONS TO ENSURE ITS QUALITY AS A BIOPHARMACEUTICAL EXCIPIENT

Size Exclusion Analysis of Tween-Containing IgG Formulations
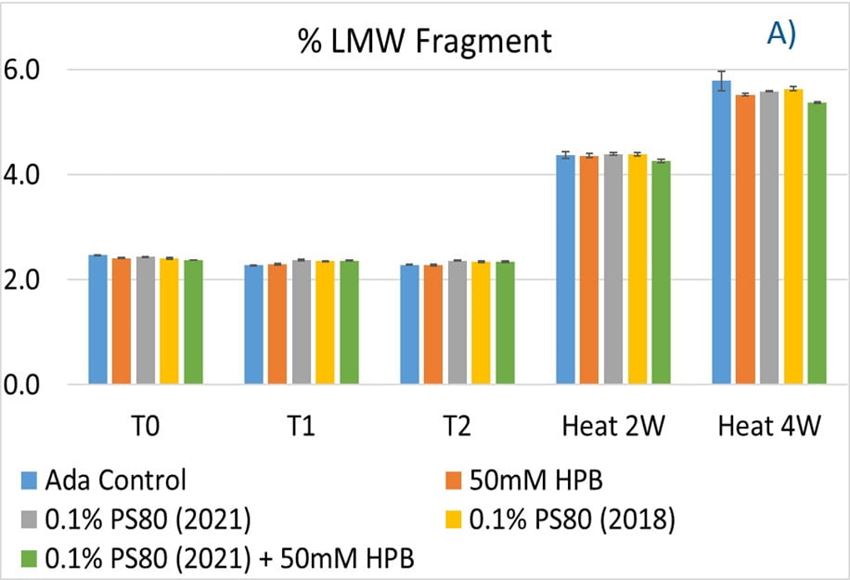
Polysorbates versus Hydroxypropyl Beta-Cyclodextrin: Comparative Study on Excipient Stability and Stabilization Benefits on Monoclonal Antibodies

Full article: Development of a stable low-dose aglycosylated antibody formulation to minimize protein loss during intravenous administration

Enhancement and analysis of Anthracene degradation by Tween 80 in LMS-HOBt