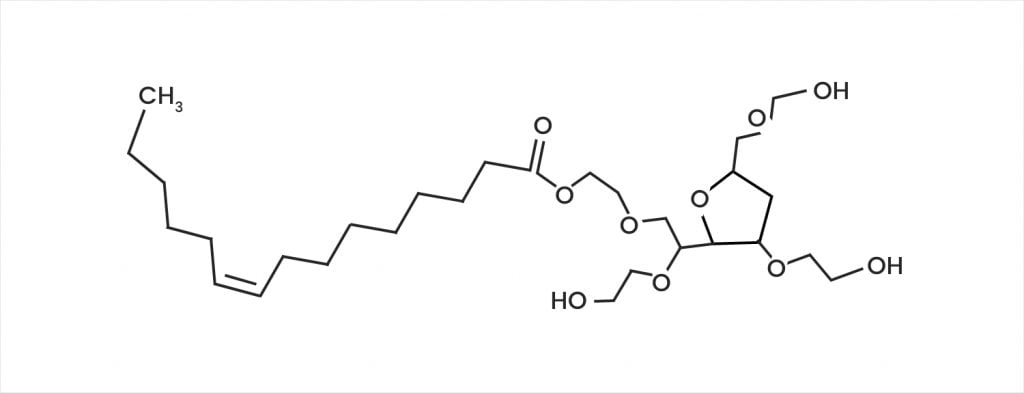
Quantitative Analysis of Polysorbate 20/80 in Protein-Based
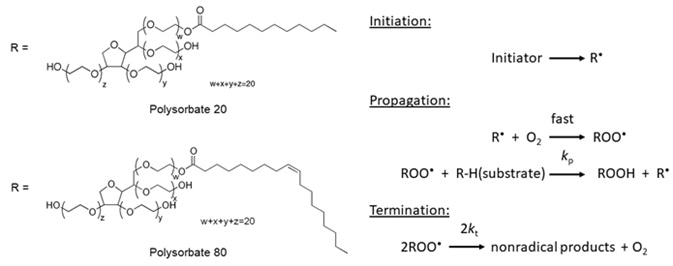
EPR Application Spotlight: Analyzing the Shelf Life of Polysorbates for the Pharmaceutical Industry
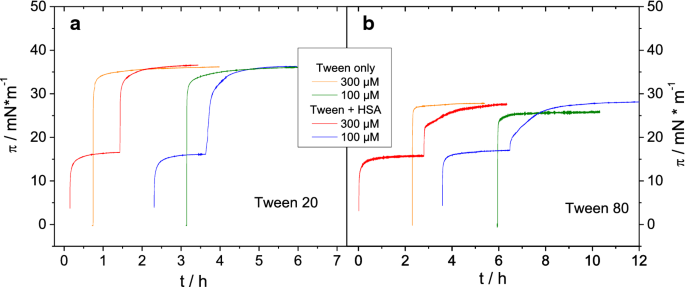
Albumin displacement at the air–water interface by Tween (Polysorbate) surfactants

Blank Wooden Sign Board Illustration Stock Vector Image Art

Polysorbate Analysis in Biopharmaceutical Applications—A Snapshot of the Analytical Toolbox

Comparing ELSD and CAD Performance on Polysorbate Quantification in Infliximab Drug Products

Fatty acid composition analysis in polysorbate 80 with high

Binding isotherms of polysorbate 20 to an immunoglobulin between 5 and

The effects of membrane filters used in biopharmaceutical processes on the concentration and composition of polysorbate 20 - Lei - 2013 - Biotechnology Progress - Wiley Online Library
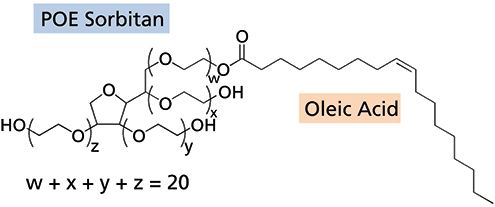
Quantification and Characterization of Polysorbate-80 in Protein Formulations

Quantitative Analysis of Polysorbate 20/80 in Protein-Based

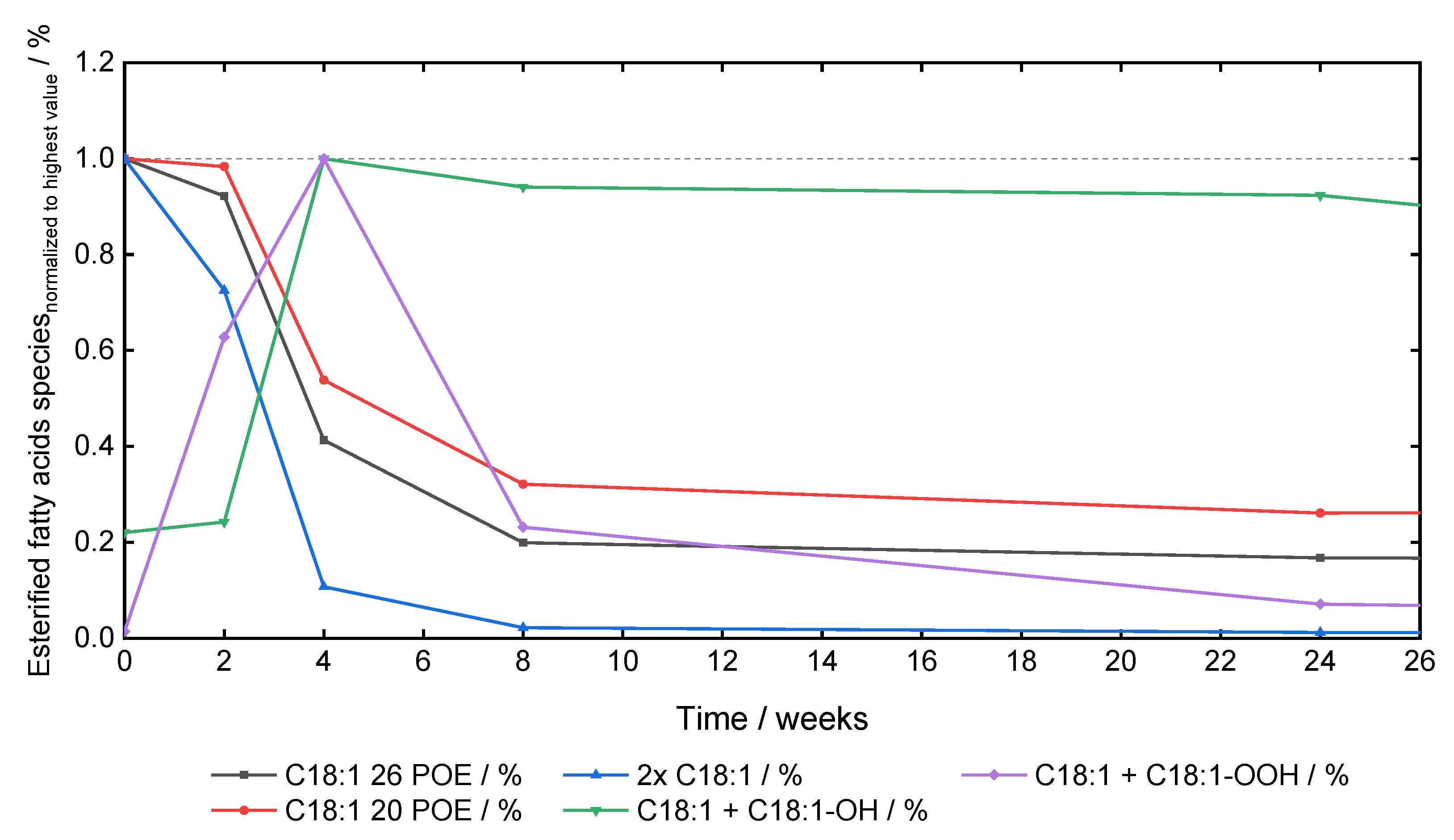
Polysorbate 20 Degradation in Biopharmaceutical Formulations: Quantification of Free Fatty Acids, Characterization of Particulates, and Insights into the Degradation Mechanism

Polysorbate 20 Degradation in Biopharmaceutical Formulations: Quantification of Free Fatty Acids, Characterization of Particulates, and Insights into the Degradation Mechanism

PDF] A Highly Sensitive Method for the Quantitation of Polysorbate 20 and 80 to Study the Compatibility between Polysorbates and m-Cresol in the Peptide Formulation
Pharmaceutics, Free Full-Text